TORONTO, ON–(Marketwired – September 27, 2016) – Titan Medical Inc. (TMD.TO) (TITXF), a medical device company focused on the design and development of a robotic surgical system for application in minimally invasive surgery (MIS), today provided an update of corporate activities and operational progress.
On September 20 2016, Titan completed a public offering of units which raised approximately US $7.2 million from US and Canadian investors. As a result of the transaction, management currently anticipates closing the third quarter ending September 30, 2016 with approximately US $8.5 million in cash.
“As a result of the recent fund raising, we have initiated active discussions with our principal development partners to implement key components of the SPORT Surgical System’s development plans within our current financial resources,” said John Hargrove, Chief Executive Officer. “At this point, we believe the resources we have on hand enable us to move forward with the advancement of human factors and usability studies of the surgical system.”
As part of the succession plan for senior management, Titan has engaged an executive search firm for the purpose of assisting in the identification and qualification of a Chief Operating Officer, whose responsibilities will include capital markets involvement, and a Vice President of Engineering to assist in product development and project management.
In addition to the operational activities, that management is focused on, the Board continues its active search to recruit a new Chairperson. We are also continuing an ongoing program of board renewal. As we achieve operational and development milestones, as well as the progress regarding our Chairperson search, we will promptly inform our shareholders.
About Titan Medical Inc.
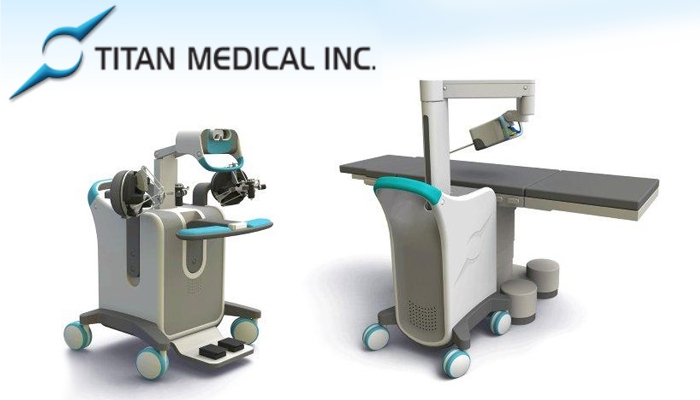
Titan Medical Inc. is a Canadian public company focused on the design and development of a robotic surgical system for application in minimally invasive surgery (“MIS”). The Company’s SPORT™ Surgical System, currently under development, includes a surgeon-controlled robotic platform that incorporates a 3D high-definition vision system and multi-articulating instruments for performing MIS procedures through a single incision. The surgical system also includes a surgeon workstation that provides a surgeon with an advanced ergonomic interface to the robotic platform for controlling the instruments and provides a 3D high-definition endoscopic view of inside a patient’s body. The SPORT™ Surgical System is designed to enable surgeons to perform a broad set of surgical procedures for general abdominal, gynecologic, and urologic indications. For more information, visit the Company’s website at www.titanmedicalinc.com.
Forward-Looking Statements
This news release contains “forward-looking statements” which reflect the current expectations of management of the Company’s future growth, results of operations, performance and business prospects and opportunities. Wherever possible, words such as “may”, “would”, “could”, “will”, “anticipate”, “believe”, “plan”, “expect”, “intend”, “estimate”, “potential for” and similar expressions have been used to identify these forward-looking statements. These statements reflect management’s current beliefs with respect to future events and are based on information currently available to management. Forward-looking statements involve significant risks, uncertainties and assumptions. Many factors could cause the Company’s actual results, performance or achievements to be materially different from any future results, performance or achievements that may be expressed or implied by such forward-looking statements, including, without limitation, those listed in the “Risk Factors” section of the Company’s Annual Information Form dated March 30, 2016 (which may be viewed at www.sedar.com). Should one or more of these risks or uncertainties materialize, or should assumptions underlying the forward looking statements prove incorrect, actual results, performance or achievements may vary materially from those expressed or implied by the forward-looking statements contained in this news release. These factors should be considered carefully, and prospective investors should not place undue reliance on the forward-looking statements. Although the forward-looking statements contained in the news release are based upon what management currently believes to be reasonable assumptions, the Company cannot assure prospective investors that actual results, performance or achievements will be consistent with these forward-looking statements.